|
hvor f er fotonens frekvens og h er Plancks konstant. I hver af disse tilstande har atomet en bestemt energi. Atommodellen var formuleret som to postulater: Atomet kan kun eksistere i nogle ganske bestemte stationære tilstande. Following the work of Ernest Rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. China produces the most tungsten out of any country in the world, producing 60,000 tonnes every year. Tungsten is used in various things, including spacecrafts, shrapnel, missiles, tools like knives, saws, mining, etc. The electrons then give off the energy in the form of a piece of light-called a photon-that they had absorbed, to fall back to a lower energy level. I 1913 opstillede Niels Bohr (1885-1962) en ny atommodel, specielt inspireret af hydrogenspektret. Tungsten has the highest melting point of all metals, clocking in at 3695 K. If the electrons are given energy (through heat, electricity, light, etc.) the electrons in an atom could absorb energy by jumping to a higher energy level, or excited state. The electrons typically have the lowest energy possible, called the ground state. When hydrogen gas is placed into a tube and electric current passed through it, the color of emitted light is pink. Thousands of new, high-quality pictures added every day. He suggested that an atomic spectrum is made by the electrons in an atom moving energy levels. The emission spectrum (or atomic spectrum) of a chemical element is the unique pattern of light obtained when the element is subjected to heat or electricity. Find Bohr Model Representation Tungsten Atom Number stock images in HD and millions of other royalty-free stock photos, illustrations and vectors in the Shutterstock collection. 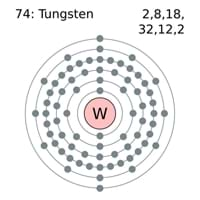 
The evidence used to support Bohr's model came from the atomic spectra. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. \): In Bohr's Model of the atom, electrons absorb energy to move to a higher level and release energy to move to lower levels. The atomic radius of Tungsten atom is 162pm (covalent radius).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
